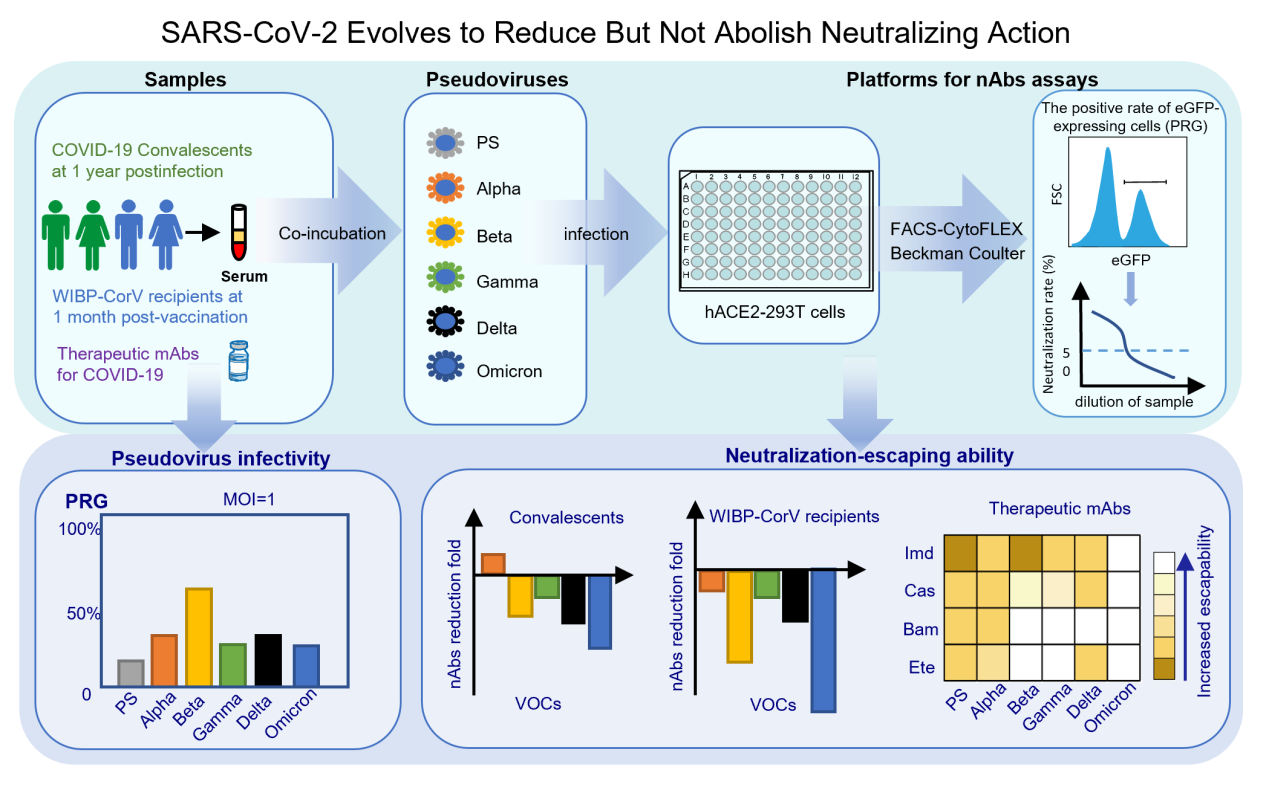
On October 11, 2022, Professor Xionglin Fan's team published a paper entitled "SARS-CoV-2 evolves to reduce but not abolish neutralizing action" in the Journal of Medical Virology. As the worldwide pandemic of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) and its disease coronavirus disease 2019 (COVID-19) has lasted more than 2 years, at least 16 vaccines and 9 monoclonal antibodies (mAbs) have been developed to conquer the pandemic based on the spike protein (S) of the SARS-CoV-2 prototype strain (PS) and approved for vaccination or treatment at unprecedented rates. Some countries and regions have put an end to lockdowns and returned to pre-pandemic normality while some scholars predicted that the pandemic is about to end. Neutralizing antibodies (nAbs) are proved to play decisive roles in preventing SARS-CoV-2 infection. However, SARS-CoV-2 continues to mutate and such variants of concern (VOCs) as Alpha, Beta, Gamma, Delta and Omicron have resulted in subsequent waves of pandemics globally. The elucidation of VOCs' mutation trends and their ability to escape neutralization is of great importance to provide a scientific basis for developing effective strategies to prevent and control the pandemic of mutant strains.

Since the outbreak of Covid-19, Prof. Xionglin Fan's team has continuously focused on the mutation of SARS-CoV-2 and its ability to escape neutralizing activity. In the paper, Lentivirus-based pseudovirus neutralization analysis platforms for SARS-CoV-2 prototype strain (PS) and VOCs were constructed, based on the spike protein of each variant and HEK 293T cell line expressing the human angiotensin-converting enzyme 2 (hACE2) receptor on the surface, and an enhanced green fluorescent protein reporter. Serum samples from 65 convalescent individuals and 20 WIBP-CorV vaccine recipients and four therapeutic monoclonal antibodies (mAbs), namely, imdevimab, casirivimab, bamlanivimab, and etesevimab, were used to evaluate the neutralization potency against the variants. The results indicate that the infection ability of SARS-CoV-2 variants may have been enhanced during the pandemic, and the virus has continued to evolve its ability to escape vaccine-induced nAb responses during the COVID-19 pandemic. Convalescent sera showed varying degrees of decreased neutralizing activity against VOCs. Compared to the PS pseudovirus control, Beta, Gamma, and Delta showed a significant ability to escape neutralization, with 2.05-, 1.43-, and 2.28-fold decreases in NT50, respectively. Omicron exhibited the most significant ability to escape the neutralizing activity of all the VOCs, with a 3.71-fold decrease in NT50 to PS pseudovirus. The study has further disclosed that the evolution of Omicron is achieved by mutating based on existing favorable mutations to gain stronger immune escape ability during the COVID-19 pandemic. It is predicted that the virus will evolve and break through the immune barrier established by infection or vaccination. Therefore, global surveillance of new viral variants, early identification and quarantine of infected individuals, and development of more effective vaccines are important measures to control the COVID-19 pandemic.
This study was supported by grants from the National Key R&D Project (2021YFC2600200), the Emergency Key Program of Guangzhou Laboratory (EKPG21-30), the Applied Basic Research Key Project of Wuhan Municipal Bureau of Science and Technology (020020601012218), and the Fundamental Research Funds for the Central Universities (HUST COVID-19 Rapid Response Call No. 2020kfyXGYJ040). PhD students Yanji Zhang, Jo-LewisBangaNdzouboukou and Xiaosong Lin are co-first authors. Professor Xionglin Fan at the Department of Pathogenic Biology, School of Basic Medicine, Tongji Medical Medicine, Huazhong University of Science and Technology, is the corresponding author.